The subventricular zone is a region situated on the outside wall of each lateral ventricle of the vertebrate brain. It is present in both the embryonic and adult brain.
In embryonic life, the subventricular zone (SVZ) refers to a secondary proliferative zone containing neural progenitor cells, which divide to produce neurons in the process of neurogenesis. The primary neural stem cells of the brain and spinal cord, termed radial glial cells, instead reside in the ventricular zone (VZ) (so-called because the VZ lines the inside of the developing ventricles).
In the developing cerebral cortex, which resides in the dorsal telencephalon, the SVZ and VZ are transient tissues that do not exist in the adult1. However, the subventricular zone of the ventral telencephalon persists throughout life.
The adult SVZ is composed of four distinct layers of variable thickness and cell density as well as cellular composition. Along with the dentate gyrus of the hippocampus, the SVZ is one of two places where neurogenesis has been found to occur in the adult mammalian brain.
Adult SVZ neurogenesis takes the form of neuroblast precursors of interneurons that migrate to the olfactory bulb through the rostral migratory stream2. The SVZ also appears to be involved in the generation of astrocytes following a brain injury.
Function Of The Subventricular Zone
The SVZ is a known site of neurogenesis and self-renewing neurons in the adult brain, serving as such due to the interacting cell types, extracellular molecules, and localized epigenetic regulation promoting such cellular proliferation. Along with the subgranular zone of the dentate gyrus, the subventricular zone serves as a source of neural stem cells (NSCs) in the process of adult neurogenesis.
It harbors the largest population of proliferating cells in the adult brain of rodents, monkeys and humans. In 2010, it was shown3 that the balance between neural stem cells and neural progenitor cells (NPCs) is maintained by an interaction between the epidermal growth factor receptor signaling pathway and the Notch signaling pathway.
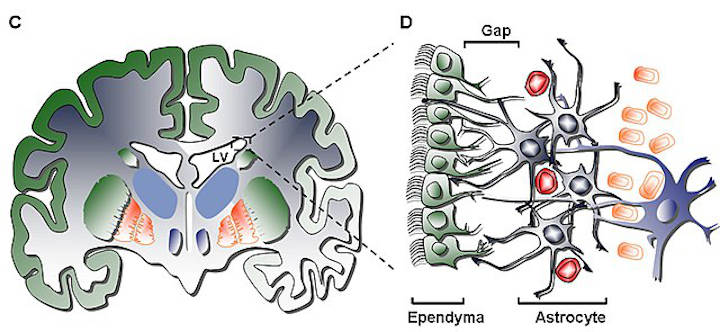
While it has yet to have been studied in-depth in the human brain, the SVZ function in the rodent brain has been, to a certain extent, examined and defined for its abilities. With such research, it has been found that the dual-functioning astrocyte is the dominant cell in the rodent SVZ; this astrocyte acts as not only a neuronal stem cell, but also as a supporting cell that promotes neurogenesis through interaction with other cells.
This function is also induced by microglia and endothelial cells that interact cooperatively with neuronal stem cells to promote neurogenesis in vitro, as well as extracellular matrix components such as tenascin-C (helps define boundaries for interaction) and Lewis X (binds growth and signaling factors to neural precursors)4.
The human SVZ is different, however, from the rodent SVZ in two distinct ways.
- The first is that the astrocytes of humans are not in close juxtaposition to the ependymal layer, rather separated by a layer lacking cell bodies
- The second is that the human SVZ lacks chains of migrating neuroblasts seen in rodent SVZ, in turn providing for a lesser number of neuronal cells in the human than the rodent5.
For this reason, while rodent SVZ proves as a valuable source of information regarding the SVZ and its structure-to-function relationship, the human model will prove significantly different.
Some current theories propose that the SVZ may also serve as a site of proliferation for brain tumor stem cells (BTSCs), which are similar to neural stem cells in their structure and ability to differentiate into neurons, astrocytes, and oligodendrocytes.
Cell Replacement After Brian Injury
Romanko et al characterized the impact of acute brain injury on the subventricular zone. Overall, the authors determined that moderate insults to the SVZ allowed for recovery while more severe injuries caused permanent damage to the region. Additionally, the neural stem cell population within the SVZ is likely responsible for this injury response.
The effects of irradiation on the subventricular zone provided for a recognition of the amount or dose of radiation that can be given is determined mostly by the tolerance of the normal cells near the tumor.
The increasing dose of radiation and age led to decrease in three cell types of the SVZ, yet repair capacity of the SVZ was observed despite the lack of white matter necrosis. This occurred likely because the SVZ was able to gradually replace the neuroglia of the brain. Chemotherapeutics were also tested for their effects on the SVZ, as they are currently used for many diseases yet lead to complications within the central nervous system.
- Rakic, P (October 2009). Evolution of the neocortex: a perspective from developmental biology. Nature Reviews Neuroscience, 10 (10): 724–35. ↩︎
- Lim, Daniel A.; Alvarez-Buylla, Arturo (2016). The Adult Ventricular–Subventricular Zone (V-SVZ) and Olfactory Bulb (OB) Neurogenesis. Cold Spring Harbor Perspectives in Biology, 8 (5): a018820. ↩︎
- Aguirre A, Rubio ME, Gallo V (September 1998). Notch and EGFR pathway interaction regulates neural stem cell number and self-renewal. Nature, 467(7313): 323–7 ↩︎
- Bernier, PJ; Vinet, J; Cossette, M; Parent, A (May 2000). Characterization of the subventricular zone of the adult human brain: evidence for the involvement of Bcl-2. Neuroscience Research, 37 (1): 67–78. ↩︎
- Quiñones-Hinojosa, A; Sanai, N; Soriano-Navarro, M; Gonzalez-Perez, O; Mirzadeh, Z; Gil-Perotin, S; Romero-Rodriguez, R; Berger, MS; Garcia-Verdugo, JM; Alvarez-Buylla, A (2006). Cellular composition and cytoarchitecture of the adult human subventricular zone: a niche of neural stem cells. The Journal of Comparative Neurology, 494 (3): 415–34. ↩︎
Last Updated on November 18, 2023