Specialized synapses in our sensory organs enable us to walk, dance, and turn our heads without experiencing vertigo or losing our balance. These synapses process signals more quickly than any other parts of the human body.
A small group of neuroscientists, physicists, and engineers from different institutions has now figured out how these synapses work. This opens the door for research that could lead to better treatments for vertigo and balance disorders, which affect up to one-third of Americans over the age of 40.
The discovery, which took more than 15 years to make, is detailed in a paper describing the functioning of “vestibular hair cell-calyx synapses,” which are located in inner ear organs and sense head position and movements in various directions.
Nonquantal Transmission
Synapses are biological intersections where neurons can communicate with one another and with other body parts. The vast majority of the hundreds of trillions of synapses in the human body communicate with one another via quantal transmission, a type of neurotransmitter-based chemical signalling that takes at least 0.5 milliseconds to complete.
Previous research had shown that a faster, “nonquantal” form of transmission occurs at vestibular hair cell-calyx synapses, which are the points at which motion-sensing vestibular hair cells meet afferent neurons that connect directly to the brain. The new study explains how these synapses work so quickly.
“Nobody fully understood how this synapse can be so fast, but we have shed light on the mystery,”
said Rob Raphael, a Rice University bioengineer who co-authored the study with the University of Illinois Chicago’s Anna Lysakowski, the University of Chicago’s Ruth Anne Eatock, current Rice graduate student Aravind Chenrayan Govindaraju and former Rice graduate student Imran Quraishi, now an assistant professor at Yale University.
One Of A Kind Structure And Function
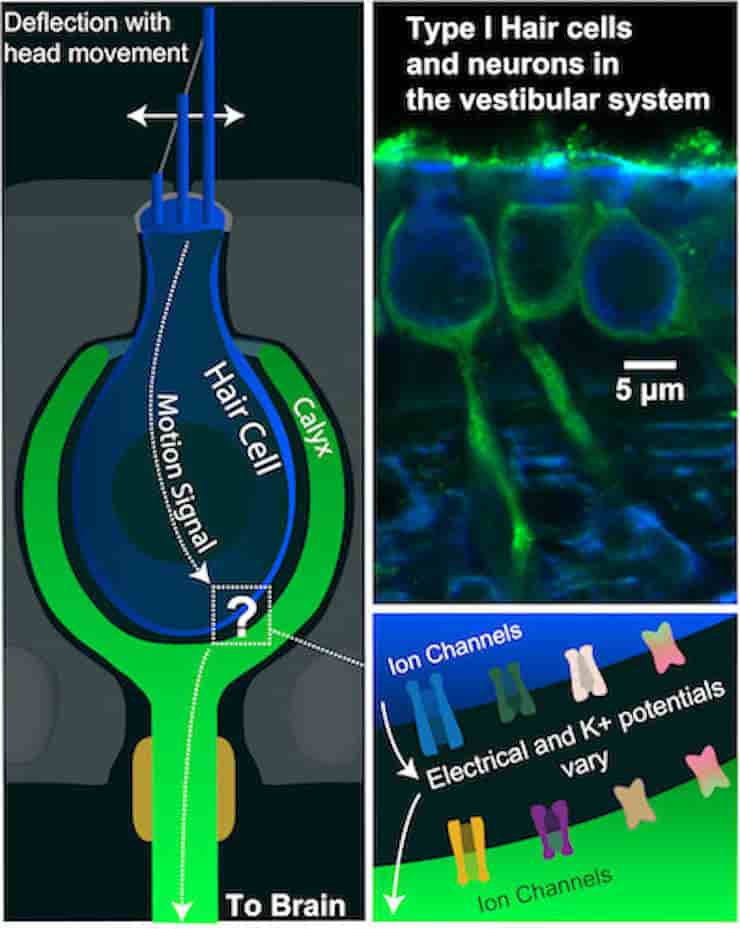
A signal-receiving neuron surrounds the end of its partner hair cell with a large cuplike structure called a calyx in each vestibular hair cell-calyx synapse. The calyx and hair cell are still separated by a tiny gap, or cleft, a few billionths of a meter wide.
“The vestibular calyx is a wonder of nature. Its large cup-shaped structure is the only one of its kind in the entire nervous system. Structure and function are intimately related, and nature obviously devoted a great deal of energy to produce this structure. We’ve been trying to figure out its special purpose for a long time,”
Lysakowski said.
The authors developed the first computational model capable of quantitatively describing the nonquantal transmission of signals across this nanoscale gap using ion channels expressed in hair cells and their associated calyces. The team was able to investigate what happens throughout the synaptic cleft, which is more extensive in vestibular synapses than other synapses, by simulating nonquantal transmission.
“The mechanism turns out to be quite subtle, with dynamic interactions giving rise to fast and slow forms of nonquantal transmission. To understand all this, we made a biophysical model of the synapse based on its detailed anatomy and physiology,”
Raphael said.
Ion Channel Model
The model tracks the flow of potassium ions through low-voltage-activated ion channels from pre-synaptic hair cells to the post-synaptic calyx to simulate the voltage response of the calyx to mechanical and electrical stimuli.
According to Raphael, the model accurately predicted changes in potassium in the synaptic cleft, providing important new insights into electrical potential changes that are responsible for the fast component of nonquantal transmission. It also demonstrated how fast and slow transmission are dependent on the close and extensive cup formed by the calyx on the hair cell, and how nonquantal transmission alone can trigger action potentials in the post-synaptic neuron.
“The key capability was the ability to predict the potassium level and electrical potential at every location within the cleft. This allowed the team to illustrate that the size and speed of nonquantal transmission depend on the novel structure of the calyx. The study demonstrates the power of engineering approaches to elucidate fundamental biological mechanisms, one of the important but sometimes overlooked goals of bioengineering research,”
Eatock said.
Closing Knowledge Gaps
Quraishi began building the model and collaborating with Eatock in the mid-2000s, when he was a graduate student in Raphael’s research group and she was on the faculty of Baylor College of Medicine in Houston’s Texas Medical Center, just a few blocks from Rice.
His first version of the model captured important synapse features, but gaps in their knowledge of the specific potassium channels and other components that comprise the model were too limited to claim it was completely accurate.
Since then, the discovery of ion channels in the calyx by Eatock, Lysakowski, and others has advanced scientific knowledge of how ionic currents move across hair cell and calyx membranes. Qurashi was relieved and delighted when Govindaraju, a Ph.D. candidate in applied physics, joined Raphael’s lab in 2018 and started working on the model because the unfinished work had weighed on him.
“By the time I started on the project, more data supported nonquantal transmission. But the mechanism, especially that of fast transmission, was unclear. Building the model has given us a better understanding of the interplay and purpose of different ion channels, the calyx structure and dynamic changes in potassium and electric potential in the synaptic cleft,”
Govindaraju said.
Calyx Structure Specialization
Raphael’s first grant was to create a model of ion transport in the inner ear.
“It is always satisfying to achieve a unified mathematical model of a complex physiological process. For the past 30 years, since the original observation of nonquantal transmission, scientists have wondered, ‘Why is this synapse so fast?’ and, ‘Is the transmission speed related to the unique calyx structure?’ We have provided answers to both questions,”
he said.
The relationship between calyx structure and function is an example of how evolution drives morphological specialization.
“A compelling argument can be made that once animals emerged from the sea and began to move on land, swing in trees and fly, there were increased demands on the vestibular system to rapidly inform the brain about the position of the head in space. And at this point the calyx appeared,”
Raphael said.
He added that the model allows for a more in-depth investigation of information processing in vestibular synapses, including research into the distinct interactions between quantal and nonquantal transmission.
He hopes the model will help those who design vestibular implants, neuroprosthetic devices that can restore function to those who have lost their balance, as well as researchers studying electrical transmission in other areas of the nervous system.
Reference:
- Aravind Chenrayan Govindaraju et al, Nonquantal transmission at the vestibular hair cell–calyx synapse: K LV currents modulate fast electrical and slow K + potentials, Proceedings of the National Academy of Sciences Vol. 120, No. 2 (2023). DOI: 10.1073/pnas.2207466120
Last Updated on January 30, 2023
