A new imaging agent which binds to tau protein and makes it visible in positron emission tomography (PET) scans has enabled scientists at Washington University School of Medicine in St. Louis to demonstrate that levels of tau are better markers of the cognitive decline characteristic of Alzheimer’s than levels of amyloid beta seen in PET scans.
“Our work and that of others has shown that elevated levels of amyloid beta are the earliest markers of developing Alzheimer’s disease,” said senior author Beau M. Ances, MD, PhD, an associate professor of neurology. “But in the earliest stages of Alzheimer’s disease, even with amyloid buildup, many patients are cognitively normal, meaning their memory and thought processes are still intact. What we suspect is that amyloid changes first and then tau, and it’s the combination of both that tips the patient from being asymptomatic to showing mild cognitive impairment.”
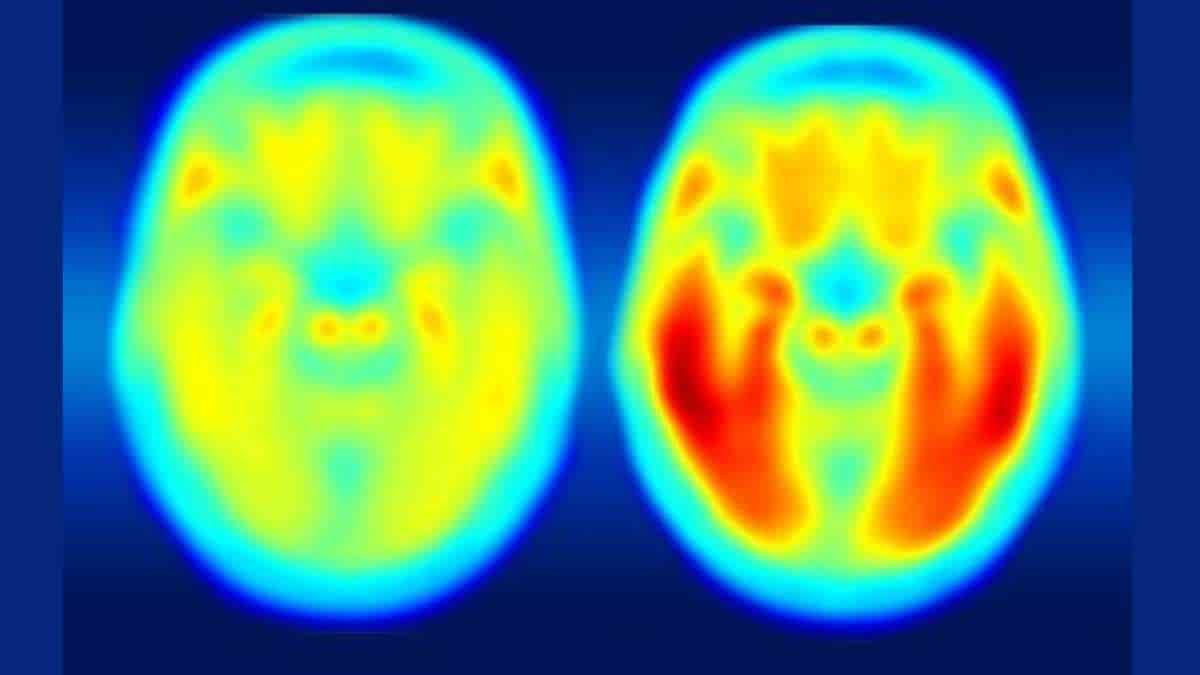
Comparing brain images of people who are cognitively normal with patients having mild Alzheimer’s disease, the researchers found that measures of tau better predict symptoms of dementia than measures of amyloid beta.
To determine degrees of cognitive impairment, some of the participants who underwent brain imaging also were assessed with the traditional clinical dementia rating scale, cerebrospinal fluid measures, and widely used pen and paper tests of memory and other brain functions.
Agent T807
The research involved 36 control participants who were cognitively normal, and 10 patients with mild Alzheimer’s disease.
Dr. Ances called for larger follow-up studies, but he said this analysis helped establish that the new tau agent, called T807, is an important tool for understanding the timeline of Alzheimer’s progression and for defining which regions of the brain are involved.
“Usually we can only diagnose patients later in the disease process, when brain function already is diminished,” he said. “We want to develop ways to make an earlier diagnosis and then design trials to test drugs against amyloid buildup and against tau buildup. While we currently cannot prevent or cure Alzheimer’s disease, delaying the onset of symptoms by 10-15 years would make a huge difference to our patients, to their families and caregivers, and to the global economy.”
It establishes a timeline, but Ances said the new tool is vital to gathering spatial information about affected brain areas. While elevated tau measured in cerebrospinal fluid has long been a marker of dementia, Ances said this type of data could not pinpoint which parts of the brain are gathering abnormal proteins.
With imaging agents for both amyloid beta and now tau available, Ances said researchers have the tools necessary to assess the effectiveness of investigational therapies against the buildup of both proteins.
T807 is approved for use in the context of clinical research trials and likely will prove to be important in imaging the brain for other types of disorders that also involve excess tau buildup, including traumatic brain injury.
Reference:
- Brier MR, Gordon B, Friedrichsen K, McCarthy J, Stern A, Christensen J, Owen C, Aldea P, Su Y, Hassenstab J, Cairns NJ, Holtzman DM, Fagan AM, Morris JC, Benzinger TLS, Ances BM. Tau and Aß imaging, CSF measures, and cognition in Alzheimer’s disease. Science Translational Medicine. May 11, 2016. DOI: 10.1126/scitranslmed.aaf2362
Image: More red color indicates more tau protein. The image on the left shows the average tau accumulation in the brains of cognitively normal people, averaged over many individuals. The image on the right shows the average amount of tau buildup in the brains of multiple people with mild Alzheimer’s symptoms. Scanning multiple individuals shows that the intensity of tau deposits correlates with the severity of cognitive dysfunction.
Credit: Matthew R. Brier
Last Updated on November 22, 2023