The inner workings of a gene network that regulates the development of spinal motor neurons in the growing chicken and mouse embryo has been discovered by researchers from University of California, Los Angeles.
The research also answers a long-standing question about why motor neurons, the nerve cells of the spinal cord that control muscle movement, form much faster than other types of neurons.
The findings have important implications for facilitating the production of motor neurons from stem cells in the lab. Stem cell-derived motor neurons could be used to repair the diseased or damaged spinal cord and to study neurodegenerative diseases such as amyotrophic lateral sclerosis (also known as Lou Gehrig’s disease) and spinal muscular atrophy.
Unprecedented Detailed View
Co-senior author Bennett Novitch, member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA, said:
“This study provides an unprecedented detailed view of how embryos produce the different cell types found in the mature spinal cord. The new experimental techniques we used, combined with collaborative efforts of biologists and computer scientists, are allowing us to gain new insight into the exquisite accuracy of embryonic development.”
During embryonic development of the spinal cord, different types of nerve cells are formed from precursor cells, called neural progenitors.
It has been known for a long time that different types of neurons form at differing speeds during development, with motor neurons forming faster than the other types of nerve cells that populate the spinal cord. However, researchers didn’t know why or how motor neurons develop this way.
Single Cell Transcriptomics
The research team used the latest molecular techniques to assess how the activity of genes change as neurons form. This was achieved using an approach called single cell transcriptional profiling, which allows the activity of all genes in individual cells to be measured simultaneously.
This gave the team snapshots of global gene activity in about 200 cells that were in the process of becoming motor neurons. To analyze these data, the team developed custom computer software to reconstruct how gene activity changes as motor neurons form.
The analysis suggested that a protein produced by a gene called Olig2, which is only expressed in the neural progenitors that create motor neurons, promotes motor neuron formation by interfering with the activity of several members of another gene family — the Hes genes. The Hes genes are known to prevent the development of neurons.
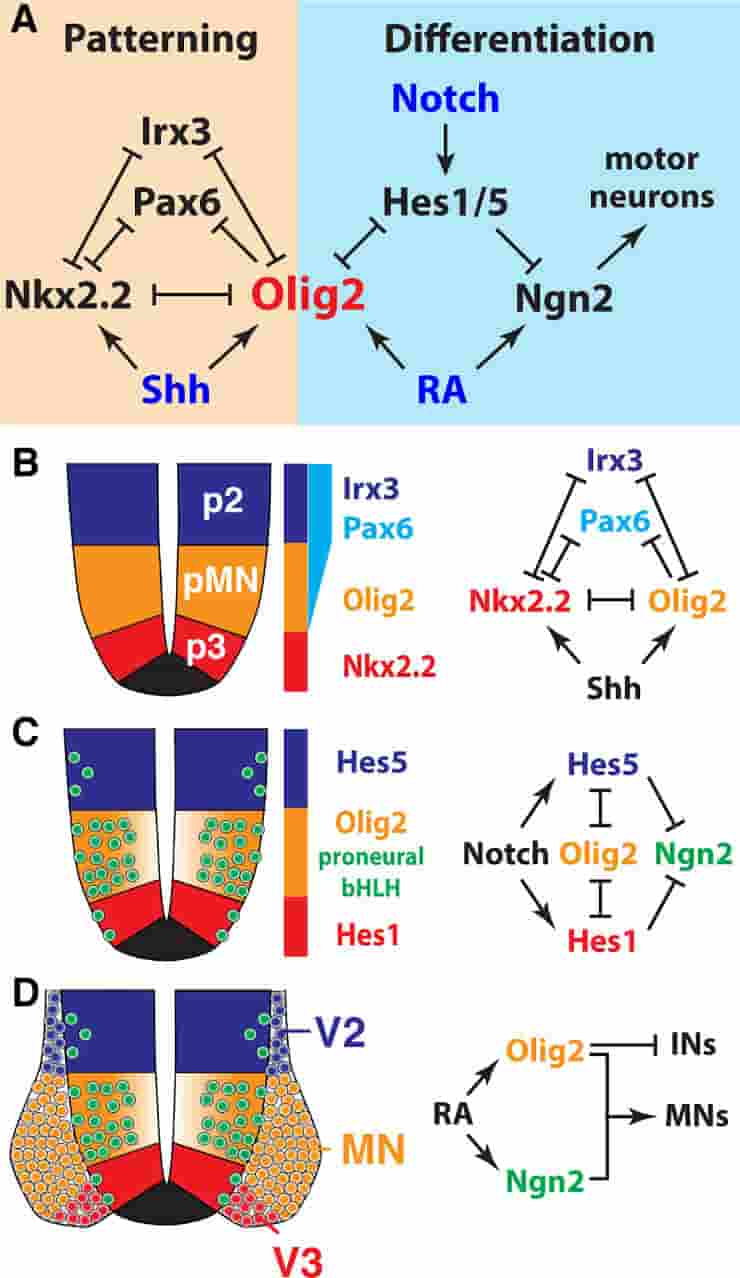
Olig2 not only acts as central organizer for dorsal-ventral patterning in the spinal cord but also controls the rate of MN differentiation through direct repression of Hes TFs. This leads to a higher levels of Ngn2 expression and, consequently, a higher rate of neuronal differentiation in the pMN domain, compared to adjacent progenitor domains.
(B) Olig2 is a core component of the Shh-controlled gene regulatory network that patterns the ventral spinal cord.
(C) Olig2-mediated down-regulation of the Notch effectors Hes1/5 relieves repression of Ngn2 in the pMN domain.
(D) Consolidated activities of Ngn2 and Olig2 cause differentiation of NPs to MNs.
Olig2 promotes differentiation of MNs through repression of alternative IN cell fates. bHLH, basic helix-loop-helix; IN, interneuron; MN, motor neuron; NP, neural progenitor; pMN, MN progenitor; p2, V2 interneuron progenitor; p3, V3 interneuron progenitor; RA, retinoic acid; Shh, sonic hedgehog; TF, transcription factor; V2, V2 interneuron; V3, V3 interneuron.
Credit: PLOS Biology/UCLA Broad Stem Cell Research Center
The researchers confirmed Olig2’s role in promoting motor neuron formation by increasing or blocking the function of Olig2 in the spinal cords of developing mouse and chicken embryos, as well as during motor neuron formation in mouse embryonic stem cells.
“During embryonic development, the nervous system is constructed following a precise blueprint, with key parts forming at precise times and places, and appropriate numbers. Our research sheds light into how this process is orchestrated specifically for motor neuron development,”
said Novitch.
Funding for the work came from Wellcome Trust, EMBO Long-term fellowship, Cancer Research UK, Whitehall Foundation, March of Dimes Foundation, NINDS, UK Medical Research Council, Human Frontiers Science Program, and People Programme (Marie Curie Actions) of the European Union’s Seventh Framework Programme.
Reference:
- Sagner A, Gaber ZB, Delile J, Kong JH, Rousso DL, Pearson CA, et al. (2018)
Olig2 and Hes regulatory dynamics during motor neuron differentiation revealed by single cell transcriptomics
PLoS Biol 16(2): e2003127.
Last Updated on October 10, 2023