Many of the same genes that lampreys have that allow for the natural repair of injured spinal cords are also active in the repair of the peripheral nervous system in mammals, including humans, according to a new study.
The findings are consistent with the possibility that the same or similar genes may be harnessed in the long term to improve spinal cord injury treatments in people.
“We found a large overlap with the hub of transcription factors that are driving regeneration in the mammalian peripheral nervous system,”
says Jennifer Morgan, director of the Marine Biological Laboratory at the University of Chicago.
Full Recovery
Lampreys are jawless, eel-like fish that shared a common ancestor with humans about 550 million years ago. The study arose from discovering that a lamprey can fully recover from a severed spinal cord without medication or other treatment.
“They can go from paralysis to full swimming behaviors in 10 to 12 weeks,”
Morgan says.
“Scientists have known for many years that the lamprey achieves spontaneous recovery from spinal cord injury, but we have not known the molecular recipe that accompanies and supports this remarkable capacity. In this study, we have determined all the genes that change during the course of recovery and now that we have that information, we can use it to test if specific pathways are actually essential to the process,”
says Ona Bloom of the Feinstein Institute for Medical Research and the Zucker School of Medicine at Hofstra/Northwell.
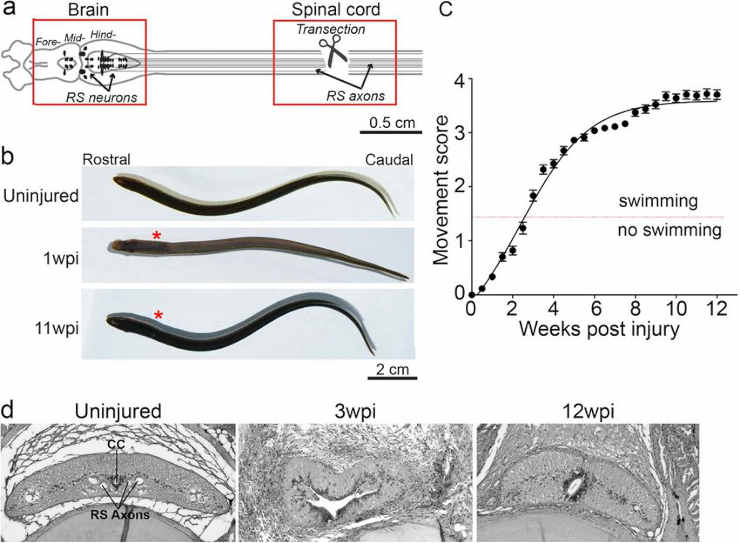
(a) Diagram of lamprey brain and spinal cord showing location of transection site and tissues collected for molecular profiling (red boxes). Somata of RS neurons (arrows) reside in the midbrain and hindbrain, and their axons (black lines) extend the length of the spinal cord.
(b) Images of uninjured lampreys and at 1wpi and 11wpi. Asterisks indicate lesion site.
(c) Quantitative scale of functional recovery (i.e. swimming): Movement scores are as described fully in18: 0-paralysis); 1-head wiggle only; 2-brief, abnormal swimming; 3-persistent swimming with abnormal body shape; 4-apparently normal swimming. Data points represent mean ± SEM (n = 27–66 animals per data point).
(d) Histological sections of lamprey spinal cord from uninjured, 3wpi, and 12wpi demonstrating cytological changes during the recovery from SCI. CC = central canal, RS axons = reticulospinal axons.
Credit: Paige E. Herman, et al CC-BY
The researchers followed the lampreys’ healing process and took samples from the brains and spinal cords at multiple points in time, from the first hours after injury until three months later when they were healed and then analyzed the material to determine which genes and signalling pathways were activated as compared to a non-injured lamprey.
Injury-induced Genetic Changes
As expected, they found many genes in the spinal cord that change over time with recovery. Further, somewhat unexpectedly, they also discovered a number of injury-induced gene expression changes in the brain.
“This reinforces the idea that the brain changes a lot after a spinal cord injury,” Morgan says. “Most people are thinking, ‘What can you do to treat the spinal cord itself?’ but our data really support the idea that there’s also a lot going on in the brain.”
They also discovered that many of the genes associated with spinal cord healing are part of the Wnt signaling pathway, a set of proteins that play a role in tissue development.
“Furthermore, when we treated the animals with a drug that inhibits the Wnt signaling pathway, the animals never recovered their ability to swim,”
Morgan says. Future research will explore why the Wnt pathway seems particularly important in the healing process.
Support for the work came from the National Institutes of Health, The Feinstein Institute for Medical Research and The Marine Biological Laboratory, including the Charles Evans Foundation Research Award, the Albert and Ellen Grass Foundation Faculty Research Award, and The Eugene and Millicent Bell Fellowship Fund in Tissue Engineering.
Reference:
- Paige E. Herman, Angelos Papatheodorou, Stephanie A. Bryant, Courtney K. M. Waterbury, Joseph R. Herdy, Anthony A. Arcese, Joseph D. Buxbaum, Jeramiah J. Smith, Jennifer R. Morgan & Ona Bloom. Highly conserved molecular pathways, including Wnt signaling, promote functional recovery from spinal cord injury in lampreys. Scientific Reportsvolume 8, Article number: 742 (2018) doi:10.1038/s41598-017-18757-1
Last Updated on February 7, 2023
