A possible way to address loss of cognitive function due to Alzheimer’s disease by targeting protein synthesis has been identified by researchers. The findings1 show that synthetic pharmaceuticals could rescue the activity of brain cells needed for memory formation.
This work is the first to show that reversing impaired protein synthesis in brains afflicted by Alzheimer’s disease through a pharmacological approach is not only feasible, but also effective,
says lead author Mauricio Martins-Oliveira, a postdoctoral researcher at New York University’s Center for Neural Science.
Impaired Synthesis
Currently, treatments for Alzheimer’s disease center on the reduction of phenomena linked to the affliction, such as amyloid plaque load, neurofibrillary tangles, and neuroinflammation. The new study suggests that the addition of a pharmaceutical normalizing protein synthesis could aid in reviving normal brain activity.
The synthesis of new proteins in the brain is essential for proper neuronal function and, notably, for memory consolidation. We and others have previously shown that impairments in brain protein synthesis contribute memory deficits in Alzheimer’s disease model mice, and that the brains of Alzheimer’s patients exhibit clear signs of impaired protein synthesis,
says co-senior author Sergio Ferreira, a professor at the Institute of Biophysics at Brazil’s Federal University of Rio de Janeiro (UFRJ).
Given the complex nature of Alzheimer’s disease, identifying and targeting abnormal molecular pathways that effectively improve cognition has been challenging. Our findings show that jump-starting protein synthesis in the brain can revive lost cognitive functions. We hope that this work can serve as a step forward in treating this devastating disease,
adds co-senior author Eric Klann, a professor in New York University’s Center for Neural Science.
Integrated Stress Response Inhibitor
The study centered on Integrated Stress Response Inhibitor (ISRIB)2, a synthetic molecule capable of boosting protein synthesis. Developed by Peter Walter at the University of California, San Francisco, ISRIB specifically targets the process of translation initiation – the translation of genetic codes – ultimately stimulating cellular protein synthesis, or the production of proteins.
Researchers wanted to find out if ISRIB could restore synaptic plasticity — the ability of the brain to change in order to learn — and memory. Previously, they had shown that translation initiation is impaired in Alzheimer’s brains, leading the team to hypothesize that ISRIB could work to re-establish some cognitive functions.
After establishing that key components of the protein synthesis machinery are depleted in the hippocampus of Alzheimer’s disease patients, the scientists concluded that protein synthesis may also be impaired.
Hippocampus Function
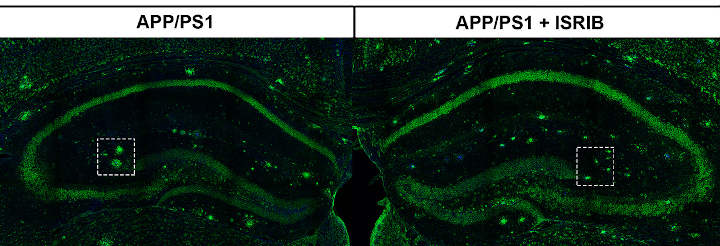
They then tested whether ISRIB could rescue the memory of mice with Alzheimer’s-like conditions by administering a series of memory tests (e.g., navigating a maze). Here, they found that ISRIB could indeed restore the memory functions in these mice as well as protein synthesis in the hippocampus.
They also examined whether ISRIB could restore the hippocampal functions and cognition in mice with fully developed Alzheimer’s-like afflictions, mimicking the disease at its more advanced stages. Similarly, their results showed that ISRIB could restore synaptic plasticity in the hippocampus, as well as memory functions.
Taken together, the researchers’ findings indicate that restoring protein synthesis, aided by synthetic molecules — in this case, ISRIB — could work to restore cognitive processes impaired by Alzheimer’s disease.
- Mauricio M. Oliveira, et al. Correction of eIF2-dependent defects in brain protein synthesis, synaptic plasticity, and memory in mouse models of Alzheimer’s disease. Science Signaling 02 Feb 2021: Vol. 14, Issue 668, eabc5429 DOI: 10.1126/scisignal.abc5429 ↩︎
- Sidrauski C, Acosta-Alvear D, Khoutorsky A, Vedantham P, Hearn BR, Li H, et al. (May 2013). Pharmacological brake-release of mRNA translation enhances cognitive memory. eLife. 2: e00498. ↩︎
Last Updated on October 3, 2022