Astrocytes are star-shaped brain cells that play an important role in maintaining the blood-brain barrier, supplying nutrients to nerve cells, and removing metabolic waste. They account for more than half of glial cells, the brain’s supporting cells that were previously thought to be nothing more than “glue” holding nerve cells together.
But this view has shifted dramatically in recent years, particularly for astrocytes. Astrocytes are now thought to play a big part in how the brain sends and receives signals. This is because their tendrils, like branches that spread out, connect nerve cells and blood vessels.
The protein Ezrin, among other important building blocks, is abundant in astrocyte tendrils. This implies that Ezrin is also important for astrocyte function during brain neuronal development. Although Ezrin has been extensively studied in vitro, there have been few in vivo studies on the role of this protein in astrocytes to date.
Now, a research team, under the direction of Prof. Helen Morrison of the Leibniz Institute on Aging—Fritz Lipmann Institute in Germany, has recently uncovered the function of Ezrin in brain development and how its absence can equip the brain to minimize damage after stress, such as a stroke.
Ezrin Functional Importance
Ezrin is mainly found in developing neurons in the developing brain and is also present in the adult brain in the peripheral astrocyte protrusions.
“But until now, there have been no comprehensive in vivo studies of its functional importance to the nervous system,”
said Morrison.
Therefore, in vivo tests were performed on mice lacking the nervous system protein Ezrin.
The cerebral cortex was the main subject of the investigations. To learn more about how Ezrin functions in the developing brain and the adult brain, special focus was given to the astrocytes and their tendrils.
Signal Processing Changes
Credit: Glia (2022). DOI: 10.1002/glia.24253
The mice’s completely normal development despite the absence of Ezrin initially surprised the researchers.
“Compared to the wild-type mice, they also showed no obvious deficits in learning or memory,”
said Dr. Stephan Schacke, who wrote his doctoral thesis on this topic.
It appears that as the brain grows, proteins that are structurally and functionally related to Ezrin take on its lost role and compensate for its loss. The modified mice only displayed different, slowed behaviour when exploring new environments, indicating modifications in neuronal signal processing.
Glutamate Metabolism
Proteome analyses and histological techniques were able to show that the absence of Ezrin affects crucial cell biological processes like glutamate metabolism. One of the most crucial excitatory neurotransmitters in the central nervous system, glutamate is essential for communicating signals between nerve cells.
The amount of glutamate released and the time until the neurotransmitter is reabsorbed, among other things, control the strength of signal transmission (and thus transmission ends). The protein GLAST, which is directly involved in glutamate reuptake, plays an important role in signal transmission.
In the absence of Ezrin, the amount of GLAST increases, which presumably speeds up glutamate reuptake. As a result, signal transmission is weakened as well as shortened. This could be one explanation for the modified mice’s slower exploratory behaviour.
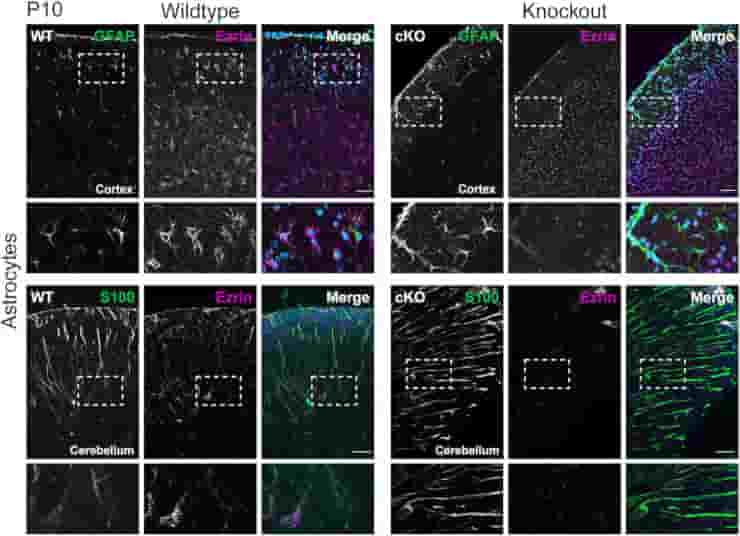
Also, GFAP, a glial filament protein also found in astrocytes and responsible for their mechanical properties, motility, and cell shape is upregulated in the absence of Ezrin. Increased levels of GFAP indicate that astrocytes have changed morphologically and adopted a “reactive status,” which is also observed in brain damage or disease.
Neurotransmitter Toxicity Mitigation
Succeeding studies revealed that, when compared to wild-type mice, the changes in astrocytes caused by Ezrin deficiency better protect these mice from stress, such as ischemic stroke, in which the brain is no longer supplied with enough blood and oxygen due to a blocked artery.
“These mice can withstand a stroke much better than their wild-type relatives because, due to the upregulation of GLAST, they have already learned to mitigate the harmfulness and toxicity of neurotransmitters, especially glutamate, which can lead to stimulus overload and neuronal death if the dose is too high,”
explained Prof. Morrison.
The study not only provides the first significant insights into the importance of the Ezrin protein for astrocyte function in our bodies, but it also points to a possible way to improve therapeutic outcome after a stroke if neuronal excitotoxicity — the injury and death of neurons caused by excessive glutamate accumulation — can be efficiently prevented. This possibility will be investigated further.
Reference:
- Schacke, S., Kirkpatrick, J., Stocksdale, A., Bauer, R., Hagel, C., Riecken, L. B., & Morrison, H. (2022). Ezrin deficiency triggers glial fibrillary acidic protein upregulation and a distinct reactive astrocyte phenotype. Glia, 70( 12), 2309– 2329.