The uncontrolled growth of benign neuroblastomas is stopped by a signal molecule produced by Schwann cells present within these tumors, new research has found.
This naturally occuring “brake” also works on malignant neuroblastoma cultures, according to scientists at St. Anna Children’s Cancer Research Institute. Together with colleagues from the Medical University of Vienna and the University of Vienna (Faculty of Chemistry), they authored the study1 describing for the first time the function of this signal molecule —not only in tumors, but also in injured nerve fibers.
Although firing a tumor with a growth factor looks contradictory at first glance, it actually makes sense in neuroblastoma.
Neuroblastoma Logic
Neuroblastoma is a tumor of the peripheral nervous system; it is the most common solid cancer in early childhood.
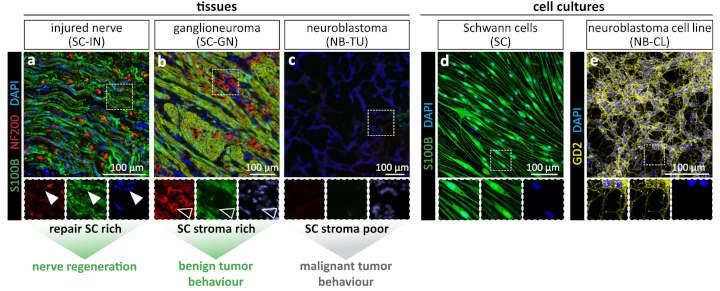
In contrast to malignant neuroblastomas, benign neuroblastomas contain many Schwann cells. These cells normally protect and repair nerve cells.
The results of the new study indicate that Schwann cells in neuroblastoma stimulate tumor cells to mature, thereby halting their unchecked growth. To accomplish this, Schwann cells produce, among other factors, a signaling molecule called epidermal growth factor like 8 (EGFL8).
Schwann Cells And EGFL8
The researchers demonstrated that EGFL8 stimulates the maturation of neuroblastoma cells.
Until recently, we only knew that this protein existed, but its function was not known. We now for the first time know where EGFL8 is produced and how it acts,
said study author Sabine Taschner-Mandl, Ph.D., head of the Tumor Biology Group at St. Anna Children’s Cancer Research Institute.
The study results also showed that high levels of EGFL8 were associated with better survival rates in neuroblastoma patients.
In cell cultures, we have demonstrated that Schwann cells as well as their secreted signaling molecules exert anti-tumor effects, even in aggressive neuroblastoma cells. Thus, we are able to exploit a process that occurs naturally in benign neuroblastomas to stop the malignant ones,
Sabine Taschner-Mandl and her colleague Tamara Weiss, Ph.D., from the Medical University of Vienna, explained.
In addition to EGFL8, other, yet uncharacterized Schwann cell molecules could also provide targets for cancer therapies in the future.
Injured Nerve Regeneration
The effects of Schwann cells may be much more extensive: the research team is currently investigating how Schwann cells manipulate immune cells in their environment.
The present study provides another significant finding: Schwann cells in benign neuroblastomas have a similar cellular status to those Schwann cells that support the healing of injured peripheral nerves.
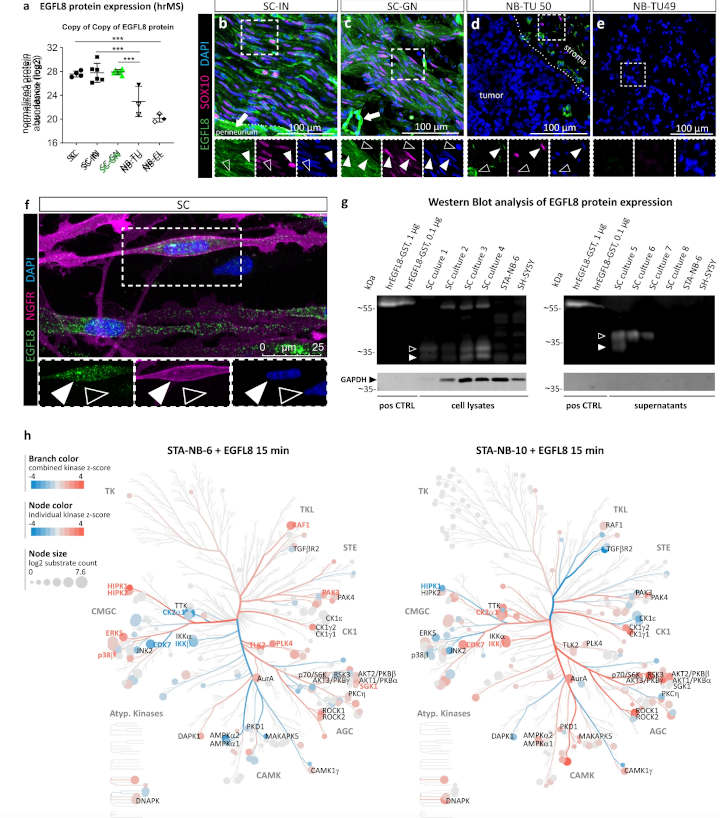
Direct comparison revealed that Schwann cells in the tumor express certain repair-associated genes and show specific repair functions.
It is amazing that we have discovered a signaling molecule that plays a role in both tumor development of benign neuroblastomas and regeneration of injured nerves. Since EGFL8 stimulates the formation of nerve cell extensions, it could be of great importance for the treatment of injured nerve fibers,
said Tamara Weiss.
It is conceivable that EGFL8 and other factors produced by Schwann cells could be applied in the treatment of nerve damage as well as aggressive neuroblastoma.
Using phosphoproteomics, we were able to decipher which signaling pathways are activated by EGFL8 in neuroblastoma cells. There are major differences compared to cells that have not been stimulated with EGFL8,
Sabine Taschner-Mandl said.
In addition to EGFL8, these downstream signaling pathways also represent potential targets for future treatments. There is still a long road to travel before these findings someday reach patients. But a firm foundation has been laid for taking the next steps.
- Weiss, T., Taschner-Mandl, S., Janker, L. et al. Schwann cell plasticity regulates neuroblastic tumor cell differentiation via epidermal growth factor-like protein 8. Nat Commun 12, 1624 (2021). ↩︎
Last Updated on October 3, 2022