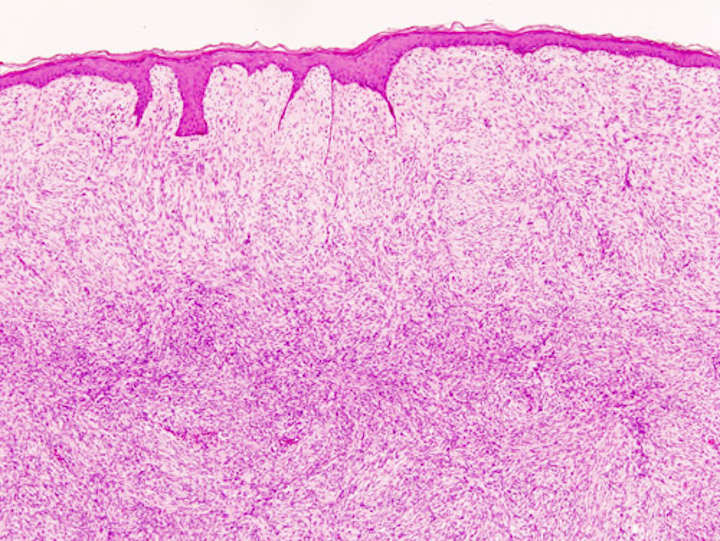
Dermatofibrosarcoma protuberans (DFSP) is a rare locally aggressive malignant cutaneous soft-tissue sarcoma. DFSP develops in the connective tissue cells in the middle layer of the skin (dermis).
Estimates of the overall occurrence of dermatofibrosarcoma protuberans in the United States are 0.8 to 4.5 cases per million persons per year. In the United States, DFSP accounts for between 1 and 6 percent of all soft tissue sarcomas and 18 percent of all cutaneous soft tissue sarcomas. In the Surveillance, Epidemiology and End Results (SEER) tumor registry from 1992 through 2004, DFSP was second only to Kaposi sarcoma.
Dermatofibrosarcoma Protuberans Diagnosis
DFSP is a malignant tumor diagnosed with a biopsy, when a portion of the tumor is removed for examination. In order to ensure that enough tissue is removed to make an accurate diagnosis, the initial biopsy of a suspected DFSP is usually done with a core needle or a surgical incision.
Clinical palpation is not entirely reliable for ascertaining the depth of a DFSP infiltration. Magnetic resonance imaging (MRI) is more sensitive, addressing the depth of the invasion on some types of DFSP, particularly large or recurring tumors1. MRI is less accurate for identifying infiltration to head and neck tumors.
Due to the rarity, initial presentation of flat plaque (skin hardening) and the slow-growing nature of DFSP, it may be months to years without a protuberance (bump).
The dissonance between the name of the neoplasm and its clinical presentations may cause a majority of patients to experience a diagnostic delay. A 2019 research study found out of 214 patients a range between less than a year to 42 years before diagnosis (median, four years) from patients noticing a symptom to diagnosis.
Currently, a majority of patients (53%) receive a misdiagnosis by health care providers. The most frequent prebiopsy clinical suspicion included cyst (101 [47.2%]), lipoma (30 [14.0%]), and scar (17 [7.9%]).
Symptoms
Dermatofibrosarcoma protuberans begins as a minor firm area of skin most commonly about to 1 to 5 cm in diameter. It can resemble a bruise, birthmark, or pimple. It is a slow-growing tumor and is usually found on the torso but can occur anywhere on the body.
About 90% of DFSPs are low-grade sarcomas. About 10% are mixed, containing a high-grade sarcomatous component (DFSP-FS); therefore, they are considered to be intermediate-grade sarcomas.
DFSPs rarely lead to a metastasis (fewer than 5% metastasize), but DFSPs can recur locally. DFSPs most often arise in patients who are in their thirties but this may be due to diagnostic delay.
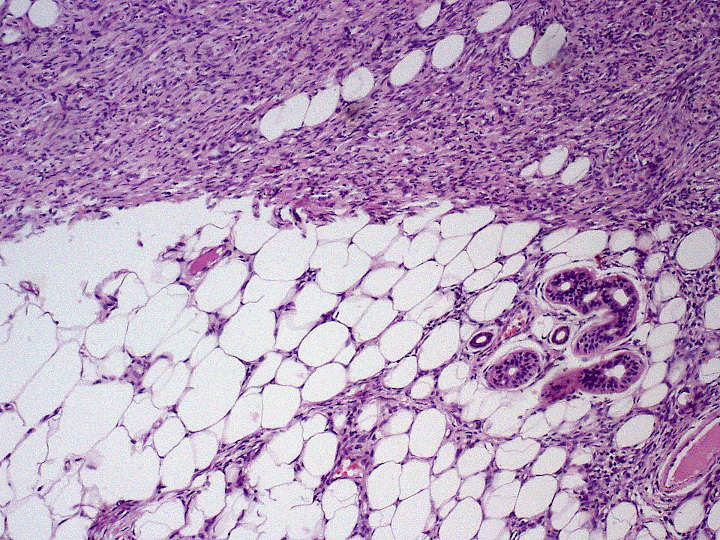
Commonly located on the chest and shoulders, the following is the site distribution of DFPS as was observed in Surveillance, Epidemiology, and End Results (SEER) database between 2000 and 2010.
- Trunk / Torso – 42%
- Lower extremity – 21%
- Upper extremity – 21%
- Head and neck – 13%
- Genitals – 1%
Treatment of DFSP
Treatment is primarily surgical, with chemotherapy and radiation therapy used if clear resection margins are not acquired.
Surgical
The type of surgical treatment chosen is dependent on the location of the DFSP occurrence and possible size.
Mohs Surgery
Mohs Micrographic Surgery (MMS) has a high cure rate and lowers the recurrence reduction of DFSP if negative resection margins are achieved.
Wide Local Excision
Wide Local Excision (WLE) was the gold standard for treating DFSP but is currently under reevaluation. Presently in the United States, WLE may be suggested after the recurrence of MMS. Larger resection margins are suggested for WLE than MMS. Recurrence rate with WLE is about 8.5% with a lower recurrence rate related to wider excision2.
Resection Margin
DFSP characteristic features are its capacity to invade surrounding tissues, to a considerable distance from the central focus of the tumor in a “tentacle-like” fashion. This fact, coupled with diagnostic delay, may lead to inadequate initial resection.
Inadequate initial treatment results in larger, deeper recurrent lesions, but these can be managed by appropriate wide excision.
Radiation Therapy
DFSP is a radioresponsive tumor, radiation therapy (RT) is not used as the first choice for treatment.
Conservative resection through MMS or WLE is attempted first. If clear margins are not achieved RT, or Chemotherapy is recommended.
Chemotherapy
DFSP was previously regarded and nonresponsive to standard chemotherapy.
In 2006 the US FDA approved (imatinib mesylate) for the treatment of DFSP. As is true for all medicinal drugs that have a name that ends in “ib,” imatinib is a small molecular pathway inhibitor; imatinib inhibits tyrosine kinase.
It may be able to induce tumor regression in patients with recurrent DFSP, unresectable DFSP, or metastatic DFSP3. There is clinical evidence that imatinib, which inhibits PDGF-receptors, may be effective for tumors positive for the t(17;22) translocation.
It has been suggested that imatinib may be a treatment for challenging, locally advanced disease and in the rare metastatic cases It was approved for use by adult patients with unresectable, recurrent and/or metastatic dermatofibrosarcoma protuberans.
Variants Of Dermatofibrosarcoma Protuberans
The World Health Organization, 2020, classified the fibrosarcomatous DFSP (DFSP-FS) variant (also termed dermatofibroma protuberans, fibrosarcomatous) of the dermatofibrosarcoma protuberans as a specific form of the intermediate (rarely metastasizing) fibroblastic and myofibroblastic tumors and other variants of this disorder as a specific form of the intermediate (locally aggressive) fibroblastic and myofibroblastic tumors.
Bednar Tumors
Bednar, or pigmented DFSP, is distinguished by the dispersal of melanin-rich dendritic cells of the skin.
It represents 1-5 percent of all DFSP occurring in people with rich in melanin pigments4.
Bednar is characterized by a dermal spindle cell proliferation like DFSP but distinguished by the additional presence of melanocytic dendritic cells. It occurs at the same rate of DFSP on fairer skin and should be considered to have the same chances of metastasis.
Myxoid DFSP
Myxoid DFSP, which has areas of myxoid degeneration in the stroma.
Giant Cell Fibroblastoma
Giant cell fibroblastoma that contains giant cells, and is also known as juvenile DFSP. Giant cell fibroblastomas are skin and soft tissue tumors that usually arise in childhood.
They are sometimes seen in association with dermatofibrosarcoma protuberans (DFSP, hybrid lesions) or may transform or recur as DFSP.
Atrophic DFSP
Atrophic DFSP resemble other benign lesions such as morphea, idiopathic atrophoderma, atrophic scar, anetoderma or lipoatrophy. It behaves like classic DFSP.
It commonly favours young to middle aged adults. It has a slow infiltrative growth and a high rate of local recurrence if not completely excised5.
Sclerosing DFSP
Sclerosing DFSP is a variant in which the cellularity is low, and the tumor consists of uniform bundles of collagen interspersed with more typical DFSP cells.
Granular cell variant is a rare type in which spindle cells are mingled with richly granular cells, the granules being lysosomal, with prominent nucleoli.
Fibrosarcomatous DFSP (DFSP-FS)
Fibrosarcomatous DFSP, a rare variant of DFSP involving greater aggression, high rates of local occurrences, and higher metastatic potential. DFSP – FS are considered to be intermediate-grade sarcomas. Although they rarely metastasize (fewer than 5 percent of cases)
- Kransdorf, M. J.; Meis-Kindblom, J. M. (August 1994). Dermatofibrosarcoma protuberans: radiologic appearance. American Journal of Roentgenology. 163 (2): 391–394. ↩︎
- Kim, Byung Jun; Kim, Hyeonwoo; Jin, Ung Sik; Minn, Kyung Won; Chang, Hak (2015). Wide Local Excision for Dermatofibrosarcoma Protuberans: A Single-Center Series of 90 Patients. BioMed Research International. 2015: 642549 ↩︎
- Rastogi, Sameer; Dhamija, Ekta; Barwad, Adarsh; Aggarwal, Aditi; Sharma, Atul; Panday, Rambha (December 2018). Advanced Dermatofibrosarcoma Protuberans Treatment With Imatinib: Experience From a Dedicated Sarcoma Medical Oncology Clinic in India. Journal of Global Oncology. 4 (4): 1–7. ↩︎
- Liszewski, Walter; Blanchette, Derek; Cunningham, Ashley M.; Miller, Daniel D. (2016-11-01). Epidemiology of Bednar tumors in the United States. Journal of the American Academy of Dermatology. 75 (5): 1064–1066. ↩︎
- Bakry, Ola; Attia, Abdalla (2012-03-27). Atrophic dermatofibrosarcoma protuberans. Journal of Dermatological Case Reports. 6 (1): 14–17. ↩︎
Last Updated on October 3, 2022